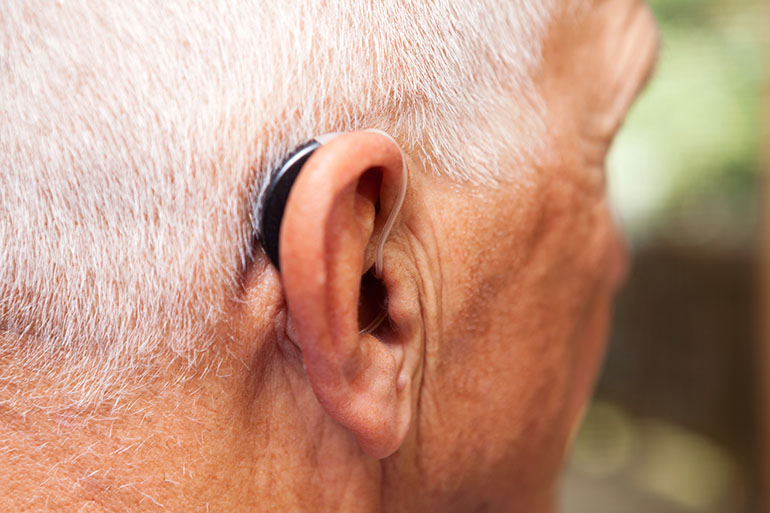
Imagine seniors walking around with stylish ear devices that amplify and clarify sound and connect wirelessly to smart phones, tablets, televisions and digital assistants such as Amazon’s Alexa or Apple’s Siri.
That day is coming, sooner than you may think.
Technology is already moving in this direction, and consumer marketers such as Samsung, Bose Corp., and Panasonic Corp. are reportedly readying new products of this kind.
They’ll be sold over the counter, to customers who will test their own hearing with cell phone apps or online programs and adjust sound parameters themselves.
The devices “will be widely used by older people,” just as earbuds are used by younger people today, predicted Richard Einhorn, a well-known composer who serves on the board of the Hearing Loss Association of America, a consumer group.
Recognizing market forces, the Food and Drug Administration is mobilizing. In December, Dr. Robert Califf, the FDA’s commissioner, said the agency planned to take “steps necessary to propose to modify our regulations to create a category of [over-the-counter] hearing aids.”
Last week, the Federal Trade Commission announced plans for a major meeting on hearing health care in April. That agency played an important role in ensuring that consumers get copies of eyeglass prescriptions so they could shop around for good deals. For the most part, that doesn’t happen with hearing aids today.
Whether agency priorities will change under the new Trump administration isn’t certain. But technology is developing rapidly under any circumstances.
Older adults with mild to moderate hearing loss, including aging baby boomers, are expected to be a prime market for a new generation of products marrying hearing aid and consumer electronics hearable technologies.
More than 40 percent of people over the age of 60 have some degree of hearing loss, mostly mild to moderate; that rises to 80 percent of people older than 80.
Yet only 20 percent of those with some degree of impairment use hearing aids because of their high cost (an average $4,700 per pair), the lack of insurance coverage (traditional Medicare doesn’t pay for hearing aids), stigma, denial and difficulty navigating the hearing health system.
Hoping to expand access, the President’s Council of Advisors on Science and Technology came out in favor of low cost, over-the-counter hearing devices in October 2015. The National Academies of Sciences, Engineering, and Medicine seconded that recommendation in a major report on hearing health care published in June.
Both organizations cite a growing body of research linked hearing loss to cognitive decline, depression, the onset of dementia, falls, poor physical functioning and social isolation. The longer people delay seeking help, research suggests, the more at risk they become.
Several recent developments are of note as consumer electronics companies, hearing aid manufacturers, audiologists, physicians, consumer advocates and regulators prepare for a surge of new hearing devices and changes in the hearing health care system:
Removing Barriers
For 40 years, the FDA has required that adults be examined by a doctor before purchasing a hearing aid or sign a waiver noting that they didn’t want to take this step. Last month, the agency eliminated that requirement for people over the age of 18.
The National Academies of Sciences’ expert panel on hearing health had noted that the rule “provides no clinically meaningful benefit” and could discourage people from seeking care. Instead of seeing a physician, adults signed the waiver 60 to 95 percent of the time.
Still, limits on access to hearing aids exist: All states restrict distribution of these devices to certified audiologists, physicians and device specialists. And some states still require medical evaluations.
Proposed Legislation
Sens. Charles Grassley, R-Iowa, and Elizabeth Warren, D-Mass., said last month that they would soon introduce new legislation endorsing over-the-counter hearing aids, sold without those restrictions.
The goal is to increase competition, lower costs and expand access to devices for people with mild to moderate hearing loss, Grassley said in a prepared statement.
Currently, six companies control nearly 98 percent of the hearing aid market in the U.S., contributing to high prices. Nearly two-thirds of people with severe hearing loss — many of them elderly — report being unable to afford the devices. The cost is generally “bundled” with professional fees for evaluation, fitting and follow-up care.
Organizations representing hearing professionals are deeply divided.
The American Speech-Language-Hearing Association, an organization representing audiologists and speech-language pathologists, doesn’t believe consumers can adequately self-diagnose hearing problems and opposes over-the-counter devices.
The American Academy of Audiology, which represents more than 12,000 audiologists, believes professionals should evaluate hearing loss but is taking a “wait and see stance” until the FDA proposes a regulatory framework, its president, Ian Windmill, said.
Another audiologist group, the Academy of Doctors of Audiology, believes the benefits of expanded access to hearing devices outweigh the risks and supports over-the-counter products.
The senators plan to introduce their legislation, which asks the FDA to issue regulations ensuring the safety and effectiveness of these devices, in this new congressional session.
“Administrations shift and legal challenges occur,” Grassley said in a statement, adding that getting the law on the books would ensure needed “certainty going forward.”
Creating Standards
One area of considerable confusion is the distinction between hearing aids and personal sound amplification products, known as PSAPs.
This is a wide category of products, ranging from cheap devices that help amplify sound to sophisticated devices that resemble hearing aids in all but their name. In some cases, companies are marketing the exact same device as a hearing aid and a PSAP, sold at different prices.
In 2009, the FDA drew a distinction between PSAPs and hearing aids based on their “intended use.” PSAPs were considered unregulated consumer electronics products for people with normal hearing who wanted to hear more sharply — for instance, during bird watching. Hearing aids were regulated and considered medical devices meant for people with hearing impairment.
But technological advances have brought the two categories closer. And it’s well understood that people with hearing loss are using PSAPs as a cheaper alternative to hearing aids.
Going forward, Stephanie Czuhajewski, executive director of the Academy of Doctors of Audiology, believes higher-end PSAPs will become over-the-counter hearing aids.
In the meantime, the Consumer Technology Association has prepared standards for PSAPs meant to make it easier for consumers to understand what they’re buying. The standards, which address issues such as maximum output, peak output and sound distortion, and are under review at the American National Standards Institute and could be published as early as next month.
“The intent is to provide a ‘Good Housekeeping Seal of Approval’ for PSAPs,” said Mead Killion, an audiologist who founded Etymotic Research, Inc., an Illinois company that manufactures hearing devices. Currently, there is no easy, standardized way to compare these devices.
We’re eager to hear from readers about questions you’d like answered, problems you’ve been having with your care and advice you need in dealing with the health care system. Visit kffhealthnews.org/columnists to submit your requests or tips.
KHN’s coverage related to aging & improving care of older adults is supported by The John A. Hartford Foundation.